
Resonance of Ketones and Esters (Carbonyls) Organic Chemistry Resonance Practice YouTube
The equivalent ressonance structures seem like the same but there are non equivalent ressonance strutures that occur when the delocalization of electrons is between qualitativity different bonds (they are different because they bond different atoms for instance a nitrogen and a carbon and two carbons) ( 6 votes) Upvote Show more.
Resonance Structures Practice Master Organic Chemistry
Practice is ESSENTIAL to mastering Organic Chemistry. You have to be able to apply the skills you are learning. First, complete the Resonance Structures Practice Quiz, and then watch this video where I go over the first three question solutions and explanations step-by-step! (Watch on YouTube : Resonance Practice.
organic chemistry Archives Organic Chemistry Made Easy by
The Resonance stabilization effect (also known as the resonance effect ), as briefly mentioned in Section 1.3, is one of the fundamental concepts of Organic Chemistry and has broad applications. The discussion of the resonance effect heavily relies on the understanding of resonance structures.
Resonance Structures Practice Master Organic Chemistry
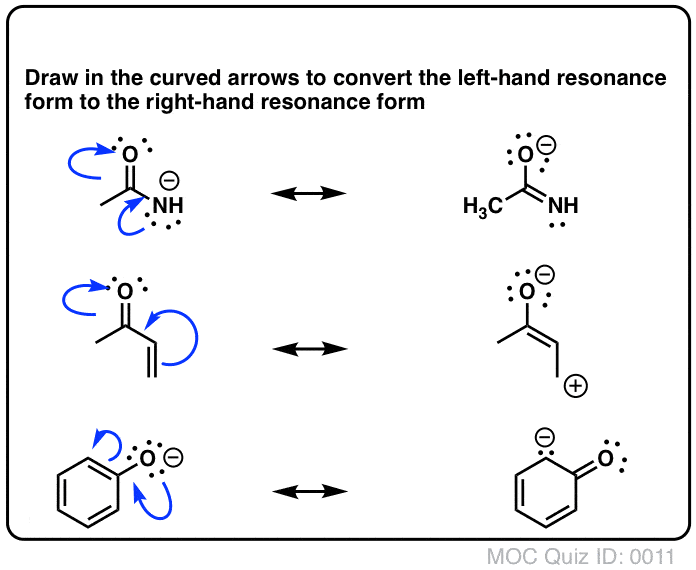
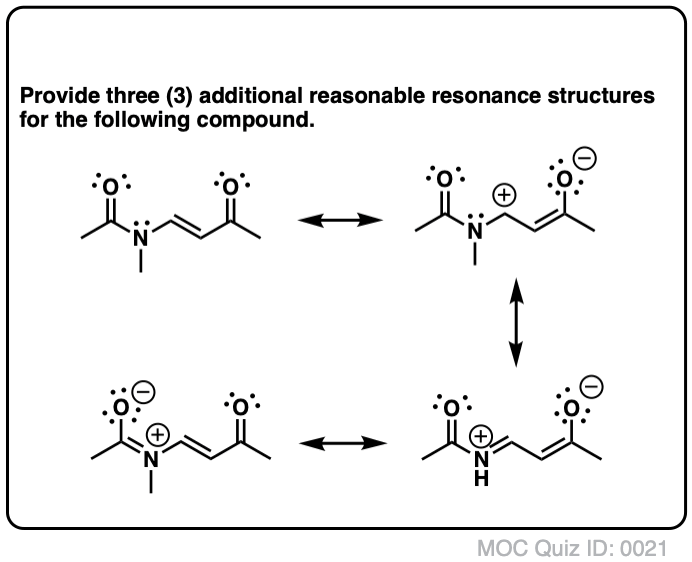
Resonance Structures Practice These questions cover all aspects of resonance in organic chemistry, from identifying resonance forms, understanding partial charges, ranking the relative importance of resonance forms, and drawing curved arrows to interconvert resonance forms. Identifying Resonance Forms Quiz#:5 Click to Flip Solution Video-A1-5

Resonance Structures Practice Problems in Organic Chemistry YouTube
1. Draw the resonance contributors that correspond to the curved, two-electron movement arrows in the resonance expressions below. 2. In each resonance expression, draw curved two-electron movement arrows on the left-side contributor that shows how we get to the right-side contributor. Be sure to include formal charges.
What is resonance? [7 rules to master it & free guide] Organic chemistry help
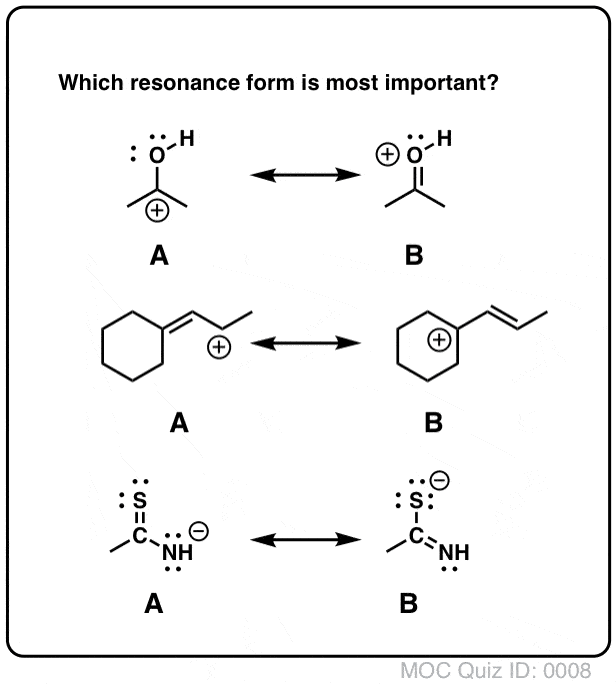
As we've seen in previous posts, four key factors that determine the importance of resonance structures in organic chemistry are: Rule #1: Minimize charges Rule #2: Full octets are favored Rule #3: How stable are the negative charges? Rule #4: How stable are the positive charges?

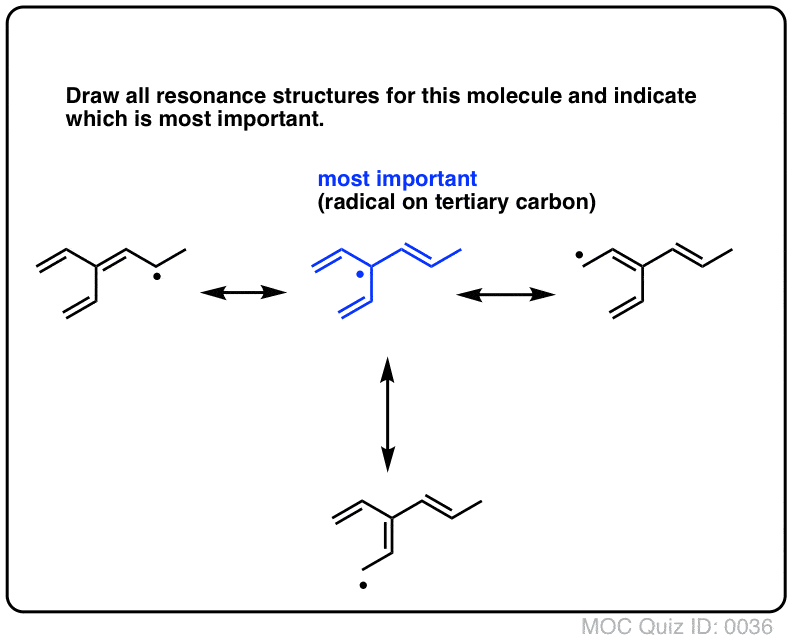
Determine which resonance structure makes the greatest contribution to the resonance hybrid. I.e
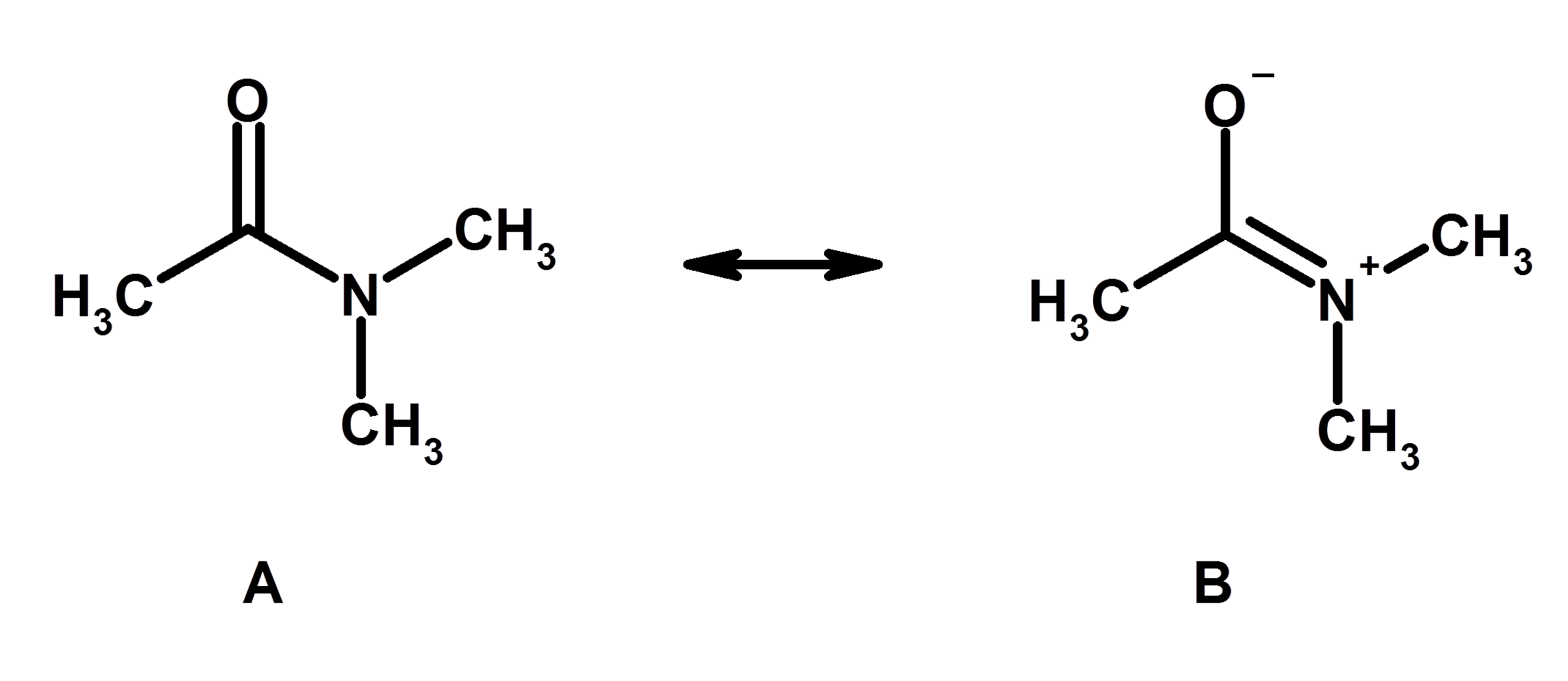
Organic Chemistry Molecular Representations Resonance Structures Practice Problems In the previous post, we talked about the resonance structures and the main rules applied to them. In summary, there are two must-follow rules when drawing resonance structures: 1) Do not exceed the octet on 2nd-row elements. 2) Do not break single bonds

6.2. Resonance Organic Chemistry 1 An open textbook
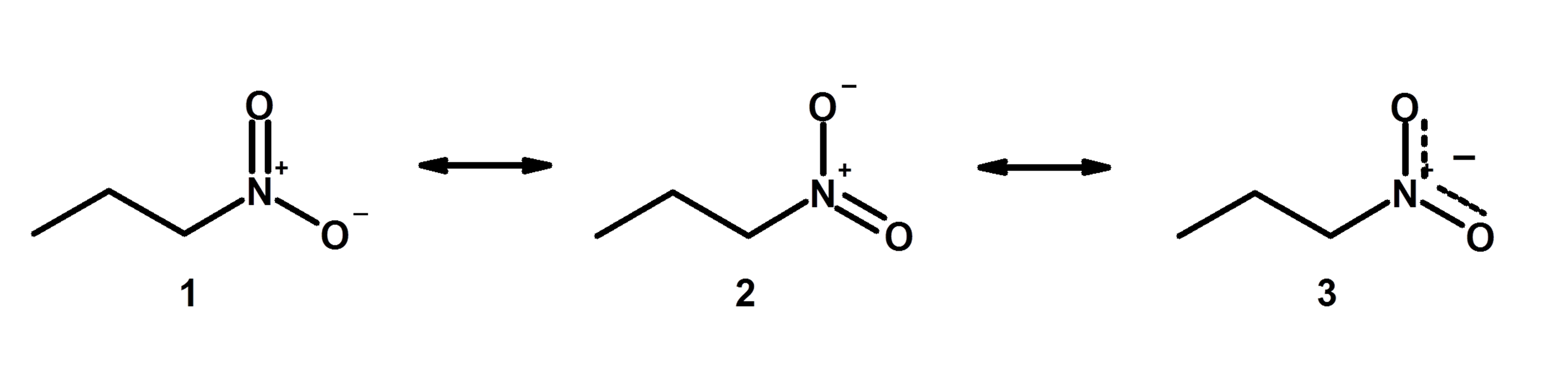
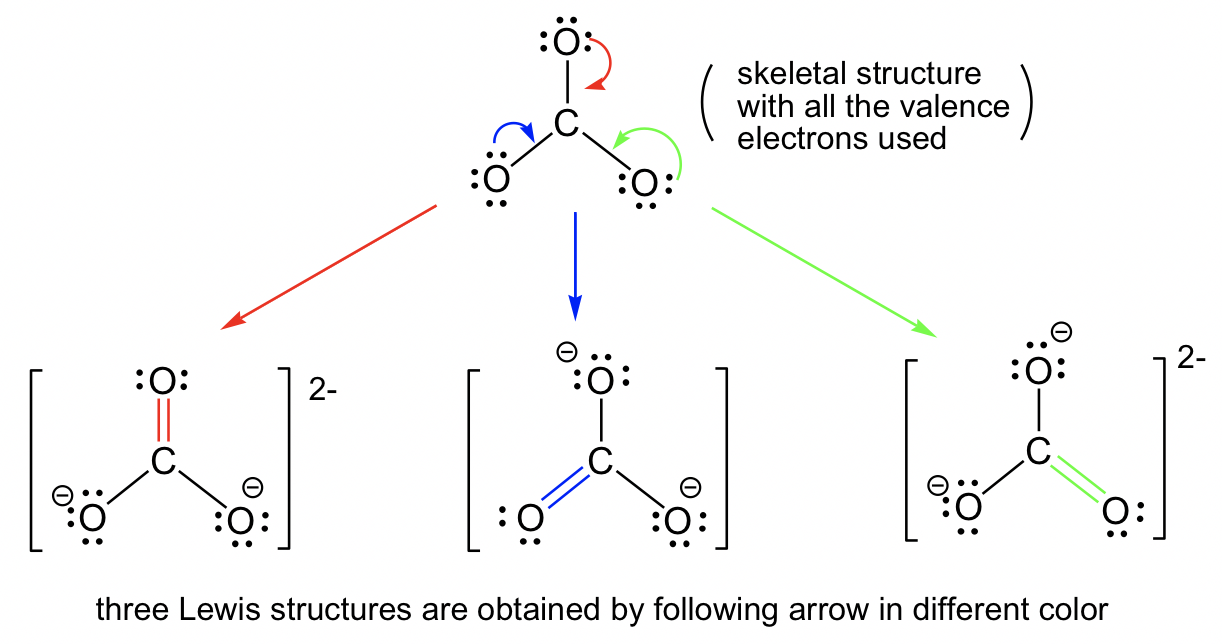
Answers to Practice Questions Chapter 1. Non-equivalent Resonance Structures. Resonance structures can also be non-equivalent. For the example of OCN -, there are three non-equivalent resonance structures, depending on how the multiple bonds are formed in Step 6 of the Lewis structure drawing procedure.. Figure 1.3e Three non-equivalent resonance structure contributors of OCN-
draw a second resonance structure for the following radical doodleArtDrawingSchool
Choose 1 answer: All of the bonds in CO A 3 A 2 − are identical in length and strength. A. All of the bonds in CO A 3 A 2 − are identical in length and strength. The bonds in CCl A 4 are more polar than the bonds in CH A 4 . B. The bonds in CCl A 4 are more polar than the bonds in CH A 4 . Both of the bonds in BeH A 2 have a bond order of 1 .

Practice drawing resonance structures organic chemistry ideas in 2022 DRAWING 99
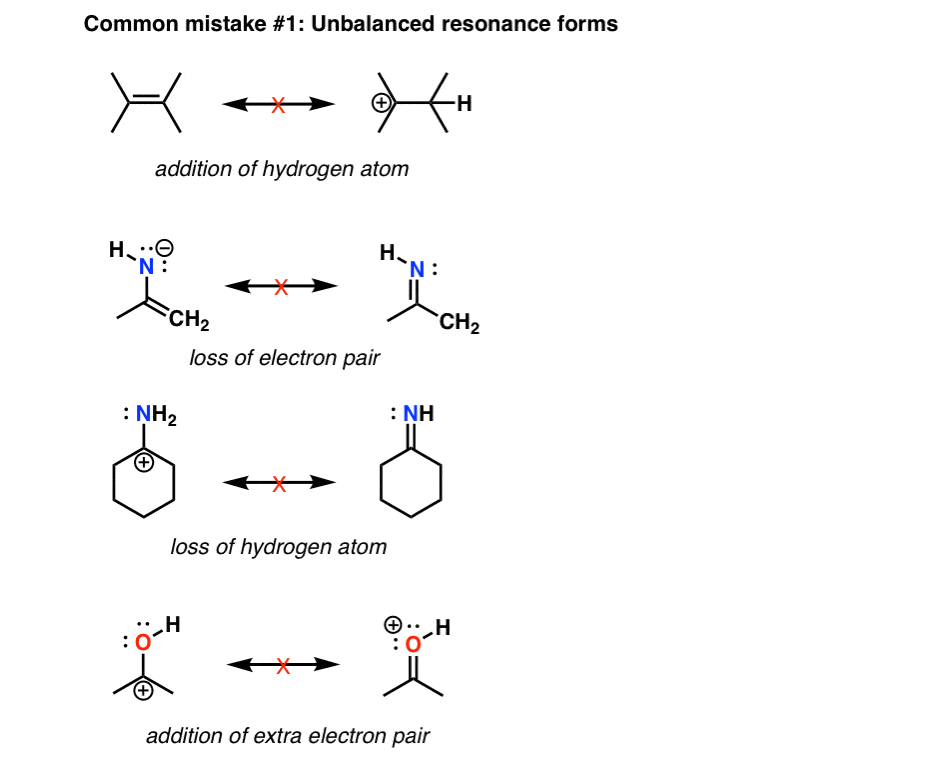
Organic chemistry (Essentials) - Class 11 Course: Organic chemistry (Essentials) - Class 11 > Unit 5 Lesson 2: Resonance Resonance structures Resonance structure patterns Resonance structures for benzene and the phenoxide anion Common mistakes when drawing resonance structures Identifying the correct resonating structure. Stability due to Resonance
draw all reasonable resonance structures for the following species jimmorrisonpaintingpawnstars
organic chemistry resonance structures practice from CLC. practice on resonance structures answers o3 for each of the following draw reasonable resonance.. organic chemistry resonance structures practice from CLC. Course. Introductory Organic Chemistry (CHEM 343) 112 Documents. Students shared 112 documents in this course.

️Resonance Worksheet Organic Chemistry Free Download Gmbar.co
1.3 Resonance Structures - Organic Chemistry I 1.3 Resonance Structures In cases in which more than one reasonable (plausible) Lewis structure can be drawn for a species, these structures are called resonance structures or resonance contributors. Resonance structures can be either equivalent or non-equivalent. Equivalent Resonance Structures

Lewis Structures in Organic Chemistry Chemistry Steps
Practice Sets Organic Chemistry II Table of Contents • Online Organic Chemistry II, Chem 360,. resonance anions more stable than anions without resonance Test 1 PS# 2: Acid Base P ractice Set. Organic(Chemistry(2((Jasperse( ( Test(1,(Alcohol(Chemistry(

How to Study the Resonance Effect in Organic Chemistry 6 Steps
Resonance structures are required throughout organic chemistry. You'll learn how to draw resonance early in orgo 1, and be tested on resonance intermediates in advanced orgo 2 mechanisms. To help you take away the guesswork I've put together a brand new series taking you through the basics, starting with the question: What IS Resonance, all the.
Resonance Structures Practice Master Organic Chemistry
Organic Chemistry Date_____ Period _____. Draw all of the reasonable resonance structures for each of the following molecules. a. f. k. p. H b. g. l. q. c. h. m. r. d. i. n. s. e. j. o. t. H2N O CN 2N O NH2 O N CN O NH2 O CN H2N O H2N NH2 O N CN H2N O O O HN HN CN O O OH. Answers to Even More Resonance Practice Problems a. b. c. H2N O H2N O.
1.3 Resonance Structures Organic Chemistry I
Organic Chemistry (Morsch et al.) 2: Polar Covalent Bonds; Acids and Bases 2.6: Drawing Resonance Forms Expand/collapse global location